Clostridia Bacteria, Dopamine Beta-Hydroxylase and Fatty Acid Beta-Oxidation Inhibition, and Their Links to Autism (part 2)
Coenzyme A (CoASH) and its influence on cellular energy metabolism - Medical Academy of Pediatric Special Needs - part 10b
On November 3rd, 2023, I posted a webinar lecture on the relationship between the gut microbiome, mucosal invasive candida, and clostridia bacteria - https://drwoeller.substack.com/p/gut-microbiome-mucosal-invasive-candida. This short lecture provides a good overview of HPHPA and its influence on dopamine. In addition, the lecture showed the relationship between these organisms and mucosal immunity, innate immunity, and the potential for autoimmune reactivity. Both of these topics will be explored in more detail in future Substack articles.
In this article, I discuss other biochemical pathways that may be altered by the presence of HPHPA, some of which are mentioned in Shaw’s article, as well as from educational content I have created for health practitioner lectures and courses.
Recap of Article #1
The following is an abbreviated list of reactions discussed in article #1 of this Substack series:
HPHPA (and other clostridia compounds, e.g., 4-cresol) inhibits DBH and its conversion of dopamine to norepinephrine.
HPHPA sequestration of free coenzyme A (CoASH) interferes with fatty acid metabolism and various enzyme complexes needed for cellular metabolism.
Depletion of free CoASH can lead to lower than normal cholesterol seen in autistic individuals.
Depletion of free CoASH can compromise palmitic acid transformation to palmitate-CoA.
Both cholesterol and palmitate-CoA are needed to activate Sonic Hedgehog (SHH).
Lack of activated SHH can compromise brain and nervous system development and maintenance.
Autism is associated with SHH problems.
To provide more depth to these points, particularly with regards to potential alterations in enzyme reactions beyond inhibition of DBH, let’s take a look at other pathways in the cell where free CoASH is needed.
Pantothenic Acid to Coenzyme A
The following image shows the relationship between coenzyme A and pantothenic acid (vitamin B5). There are multiple enzyme reactions needed to make the conversion, including PANK and PPCDC. Both of these enzymes can be affected by genetic variations, along with the others.
Notice that 4-phosphopantethine, which is past both previously mentioned enzymes, is closest to the eventual production of coenzyme-A. Pantethine (a disulphide of pantetheine) is a supplement alternative to pantothenic acid which bypasses both PANK and PPCDC.
Image 1 - the biochemical steps between pantothenic acid (vitamin B5) and coenzyme A (CoASH) takes multiple conversions. Pantethine may be a good option over pantothenic acid for CoASH support.
The next image is a screen shot of a relevant gene mutation for PPCDC (phosphopantothenoylcysteine decarboxylase). This image comes from a genomic profile of an autistic young adult.
Coenzyme A Linked To Various Enzyme Complexes
There are a number of important enzyme complexes that are critical for energy metabolism. Therefore, a reduction in free CoASH can alter various pathways involved in adenosine triphosphate (ATP) production.
Pyruvate dehydrogenase complex (PDC) - this enzyme complex is linked to the end point of glycolysis (which occurs in the cytosol) in the conversion of pyruvic acid, aka pyruvate, to acetyl-CoA. Pyruvate is converted to acetyl-CoA within the mitochondria, and participates as a cofactor for citrate synthase production of citric acid.
The PDC is a complex of proteins dependent on different nutrients. Thiamine (vitamin B1) is the rate-limiting vitamin for PDC function, as well as vitamins B2, B3, B5 (linked to CoASH), and lipoic acid.
This grouping of nutrients plays the same role in alpha-ketoglutarate dehydrogenase and branched-chain alpha-ketoacid dehydrogenase complexes.
Image 3 - pyruvate to acetyl-coA conversion via the pyruvate dehydrogenase complex. CoA-SH (coenzyme A), FAD (flavin adenine dinucleotide, B2), NAD (nicotinamide adenine dinucleotide, B3), TPP (thiamin pyrophosphate, B1), lipoate (lipoic acid).
Alpha-ketoglutarate dehydrogenase complex, aka oxoglutarate dehydrogenase complex (OGDC)
Image 4 - the citric acid cycle, aka Krebs cycle, is involved in taking acetyl-CoA and converting oxalacetate to citrate in the cycles first reaction. OGDC converts NAD+ to NADH through the enzyme complex and associated nutrients, the same as those for PDC.
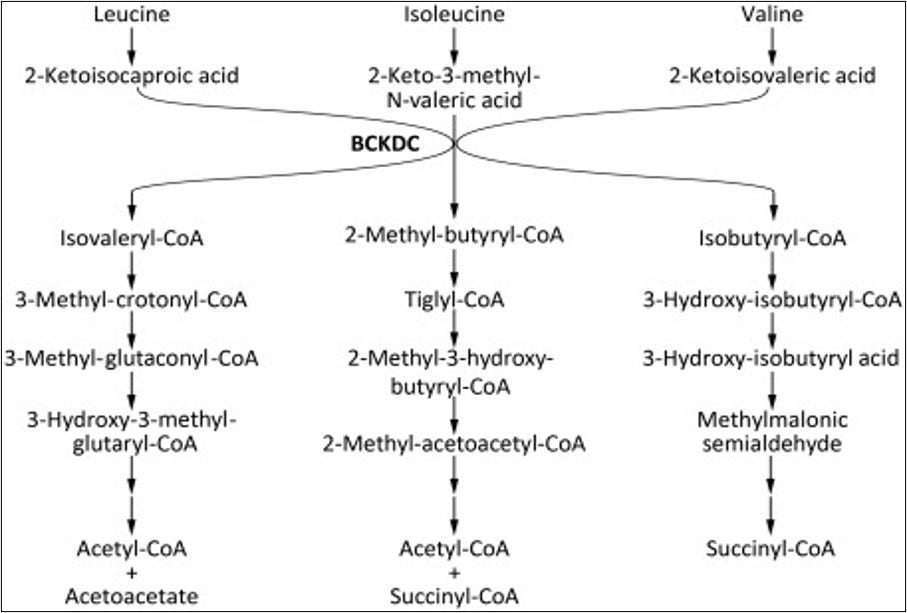
Branched-chain alpha-ketoacid dehydrogenase complex (BCKDC)
Image 5 - branched-chain amino acids of leucine, isoleucine, and valine find their way into pathways linked to acetyl-CoA and succinyl-CoA production via the BCKDC. Valine and isoleucine are linked to succinyl-CoA as another entry point into the Krebs cycle. Source: https://www.sciencedirect.com/topics/medicine-and-dentistry/isovaleryl-coa
It’s important to keep in mind that these biochemical reactions are happening within each cell that contain mitochondria (all them except red blood cells) in their production of ATP. With CoASH being a critical component in mitochondrial production of ATP, it means the presence of HPHPA can lead to poor mitochondrial activity. In autism, mitochondrial dysfunction is prevalent and can be a major factor in their overall clinical presentation.
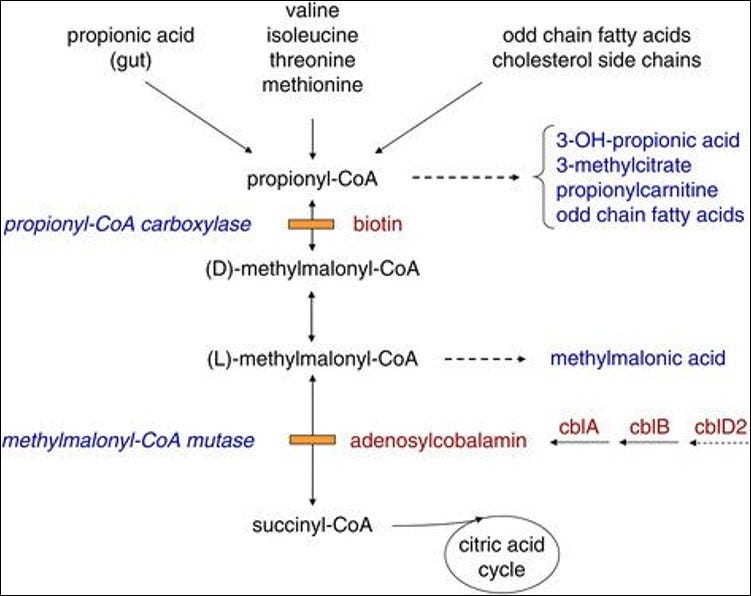
The Role of Propionyl-CoA
Another important pathway for mitochondrial metabolism is through propionyl-CoA, and the conversion of specific amino acids, odd chain fatty acids, cholesterol, and propionic acid to succinyl-CoA. This pathway too is dependent, in part, on the presence of CoASH.
Image 6 - propionyl-CoA is the conversion point for propionic acid, odd chain fatty acids, cholesterol side chains, and isoleucine and valine. These compounds get converted into succinyl-CoA for entry into the Krebs cycle.
This pathway of succinyl-CoA production is also dependent on biotin and vitamin B12. A deficiency of B12, in the form of adenosylcobalamin, can lead to methylmalonic aciduria (MMA).
The Big Picture
The final image is a metabolism summary showing interconnections between various proteins (amino acids), carbohydrates, and fats for either acetyl-CoA or succinyl-CoA production.
Image 7 - amino acids, carbohydrates, and fatty acids through various conversion sequences can find their way into the mitochondria for ATP production. Many of these sequences are dependent on CoASH.
Conclusion
What has not been discussed at this point is fatty acid metabolism, specifically beta-oxidation which plays an important role in cellular metabolism of fats. This is critical information to understand because the presence of HPHPA from clostridia appears to disrupt this biochemical process too. This topic is the focus of part #3.