Plasmalogens, Cell Membrane Function, and Implications in Chronic Inflammatory Diseases (part 1)
What are plasmalogens, what do they do, and why is it important to know about them?
In this two-part article series I am going to describe the role of plasmalogens with regards to cell membrane function, which also effects intracellular activity, and their connection to aging, inflammation, and chronic neurodegenerative disorders such as Alzheimer’s and Parkinson’s diseases. Along the way, you will also see how mitochondrial damage can impact plasmalogen activity (and vice versa), and why oxidative damage effecting a cell can have broad reaching influences on cellular function.
This article series draws on information from two papers from the journal Frontiers which were published in August 2020 (Advances in the Biosynthetic Pathways and Application Potential of Plasmalogens in Medicine) and October 2021 (Plasmalogens and Chronic Inflammatory Diseases). For a more detailed information related to specific topics such as plasmalogen chemistry, various disease associations, and literature references make sure to access the two articles.
Introduction
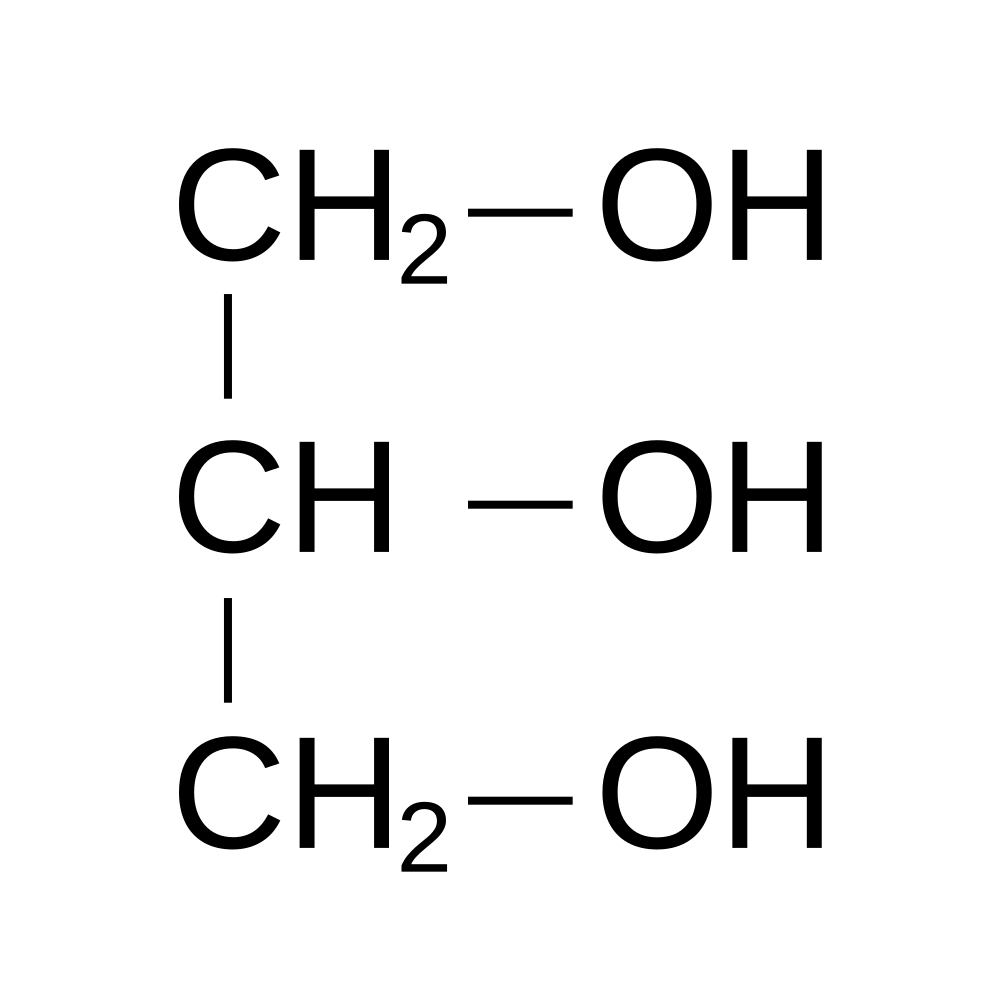
Plasmalogens are a specific class of glycerophospholipids that contain a unique chemical characteristic that sets them apart from other cell membrane phospholipids. In short, and to be described more in-depth later, plasmalogens contain a enyl-ether (vinyl-ether) bond at sn-1 and an ester bond at sn-2 of the glycerol backbone (image 1 below). All glycerolipids contain a glycerol backbone.
From a functional standpoint, plasmalogens are necessary for maintaining rigidity of the membrane for overall shape and curvature. Within the cell plasmalogens are concentrated in certain organelles that contain many folds with highly curved membranes such as the endoplasmic reticulum, Golgi bodies, and mitochondria (particularly the inner mitochondrial membrane, IMM). The plasmalogen form of cardiolipin, found in the IMM, is densely packed around electron transport chain complexes. Plasmalogens have some antioxidant capacity within the membrane, and are mportant for stabilizing membrane domains around cellular receptors which are necessary for signal transduction.
Image 1 - this chemical structure is a glycerol compound that contains 3 carbons, 3 oxygen, and 8 hydrogen atoms. The -OH (hydroxyl) groups release their hydrogen atom with subsequent bonding to various fatty acids (see next image). Source: https://byjus.com/chemistry/glycerol-formula/.
Image 2 - the glycerol is a backbone structure (b) for the binding of fatty acid (a) to form a triglyceride, aka triacylglycerol (c ). The R designation indicates a type of chemical chain or functional group which can be different for different types of glycerolipids. Notice with (c ) there is the glycerol backbone on the left side of the chemical, and a fatty acid (a) attached to each oxygen.
The plasmalogen (image 3 below) configuration allows for its unique role in cell membrane function, and a deficiency is associated with various neurodegenerative and metabolic disorders such as Alzheimer’s disease and cardiovascular illness, respectively. As mentioned previously, plasmalogen reduction is seen in aging too.
Image 3 - the chemical on the left is a glycerophospholipid, and the one of the right a plasmalogen. As an example, the sn-2 and sn-3 carbons and side chain containing a fatty acid and phospholipid, respectively are the same for both compounds. However, its important to remember that there are various types of glycerophospholipids and plasmalogens so the fatty acids attached to the glycerol backbone can be different. Notice too the plasmalogen contains a vinyl-ether (-OH-CH=CH-) linked chain (highlighted in red) at sn-1. Source: https://www.frontiersin.org/articles/10.3389/fcell.2020.00765/full.
Genetic Diseases
There are genetic diseases linked to peroxisome dysfunction and subsequent biosynthesis problems which effect the formation of plasmalogen precursors. A few examples of peroxisome disorders are Zellweger’s Syndrome (ZS) and Rhizomelic Chrondrodysplasia Punctata (RCDP), both rare autosomal recessive conditions characterized by defective plasmalogen biosynthesis. ZS leads to a devastating lack of plasmalogens effecting the brain, kidneys, liver, muscle and heart, while RCDP has varying types in its severity. Even though the mechanisms for reduced plasmalogens is different in ZS and RCDP, the end result is severe organ dysfunction.
Biosynthesis of Plasmalogens
The chemical process of plasmalogens is complex and requires the activity for two cellular organelles called peroxisomes and endoplasmic reticulum (ER). Peroxisomes are oxidative organelles often using molecular oxygen in co-substrate reactions for the production of hydrogen peroxide (H2O2). The H2O2 is involved in reduction of reactive oxygen species activity. Peroxisomes are also involved in many other cellular activities, including very long chain fatty acid catabolism for plasmalogen formation. The ER plays a role as a transportation system within the cell, as well as other functions such as protein folding and synthesis of lipids and cholesterol. The next image shows the biochemical relationship of plasmalogen formation from the activities of the peroxisome and endoplasmic reticulum.
Image 4 - peroxisomes connect with the endoplasmic reticulum (ER) in the formation of plasmalogens (plasmenylethanolamine and plasmenylcholine) through the peroxisome end-product 1-O-alkyl-DHAP. The Far1 is an important enzyme that is regulated through negative feedback by cellular plasmalogen levels.
Distribution of Plasmalogens
In most mammalian membranes, plasmalogens make up approximately 20% of the total phospholipid content. The brain has a high concentration of plasmalogens, while the liver has the lowest. Different types of plasmalogens are found in various organs too with the heart and smooth muscle being enriched with choline-plasmalogens (aka plasmenylcholine), and other organs like the brain, including the myelin shealth which surrounds neurons, having high amounts of ethanolamine-plasmalogens (aka plasmenylethanolamine).
Chemical Structure and Properties
As stated previously, plasmalogens have a unique chemical structure at the sn-1 carbon. The linkage at this position contains an ether bond as opposed to an ester bond (image 5) which is typical of glycerophospholipids.
Image 4 - this image shows an ester bond which contains a carbon double bonded to oxygen, single bonded to another oxygen, and single bonded to another functional group or side chain (R). They participate in hydrogen bonding (as a hydrogen-bond acceptor) which conveys some water solubility. They can be more volatile chemically. Source: https://www.differencebetween.com/difference-between-ester-and-vs-ether/
Image 5 - this image of an ether contains an oxygen single bonded to two functional functional groups or side chains (R, R’). Properties of ethers are described as increased tensile strength and less water soluble compared to esters. Ethers can react with oxygen to form peroxides. Source: https://www.differencebetween.com/difference-between-ester-and-vs-ether/
The enyl-ether linkage of sn-1 of plasmalogens are more hydrophobic (water repelling), more acid reactive, more oxidation labile, and less involved in hydrogen bonding. The sn-1 alkyl-chain are typically saturated or monounsaturated fatty acids around 16 to 18 carbons in length. These properties provide plasmalogens at the sn-1 position, along with their acyl-chain neighbor at sn-2, which are typically longer chain polyunsaturated fatty acids, i.e., docosahexaenoic acid (DHA) or arachidonic acid (AA), to have specific relational characteristics:
Improved motional freedom within the cell membrane
Increased membrane conformation and dynamics of the acyl-chain at sn-2
Closer packing of the proximal regions of the acyl-chain, particularly in choline-plasmalogens. As an example, this allows for increased curvature stabilization and maintenance of negative charge distribution.
A more rigid membrane bilayer which is necessary for lipid-raft domains (image 6)
Image 6 - a lipid raft is an area of the cell membrane dominated by glycerophospholipids, particularly plasmalogens, which provide stability for cell receptors. Source: https://www.researchgate.net/figure/Lipid-raft-associated-proteins-are-differentially-expressed-between-ADA-and-ADA-A_fig4_343027124.
Biological Properties
The presence of plasmalogens in the cell membrane play a role in a variety of biological functions. The following is a short-list of these functions as outlined in the two Frontier journal articles mentioned earlier:
Increase resistance to oxidative stress. This is particularly important in the brain and nervous system, but any organ system replete with plasmalogens.
Help in free radical scavenging of both reactive oxygen species (ROS) and reactive nitrogen species (RNS). This ROS/RNS scavenger ability is attributed to the oxidation-labile enyl-ether bond.
Decrease oxidative degradation of polyunsaturated fatty acids (PUFAs) with a similar efficacy of vitamin E.
Terminate lipid peroxidation. This occurs because plasmalogens oxidation byproducts are unable to further propagate oxidative reactions.
Improves endothelial cells during hypoxia
Improves signal transduction mechanism by stabilizing lipid rafts. There is some recognition that plasmalogens may have their own direct role in cellular transduction signalling.
Improve extracellular and intracellular vesicle trafficking, particularly at the neuron synapse.
Conclusion
The chemical structure, physical, and biological properties of plasmalogens place these unique glycerophospholipids as a central compound of importance for cell membrane function and maintenance. As we can see problems in plasmalogen production has profound negative consequences on health and function of various organ systems.
In part 2 of this article series, I will outline other unique characteristics about plasmalogens with regards to health and disease. Chronic inflammation is linked to many metabolic and neurodegenerative disorders, and plasmalogen maintenance is critical in preventing again tissue damage from dysregulated inflammation. However, plasmalogens themselves may be involved in inflammation too with at least one proposed role as a potential reservoir of AA and DHA.