Clostridia Bacteria, Dopamine Beta-Hydroxylase and Fatty Acid Beta-Oxidation Inhibition, and Their Links to Autism (part 3)
Review of beta-oxidation fatty acid metabolism
The main theme of Bill Shaw, PhD’s article is the various mechanisms in how HPHPA from clostridia bacteria effect cellular metabolism. A significant problem with HPHPA is its inhibition of dopamine beta-hydroxylase discussed in previous articles. Another concern for HPHPA is its potential to sequester free coenzyme (CoASH) and disrupt beta-oxidation (ß-oxidation) of certain fatty acids. For a more detailed discussion about the suspected process in how HPHPA leads to lowering of free CoASH, make sure to access Shaw’s paper.
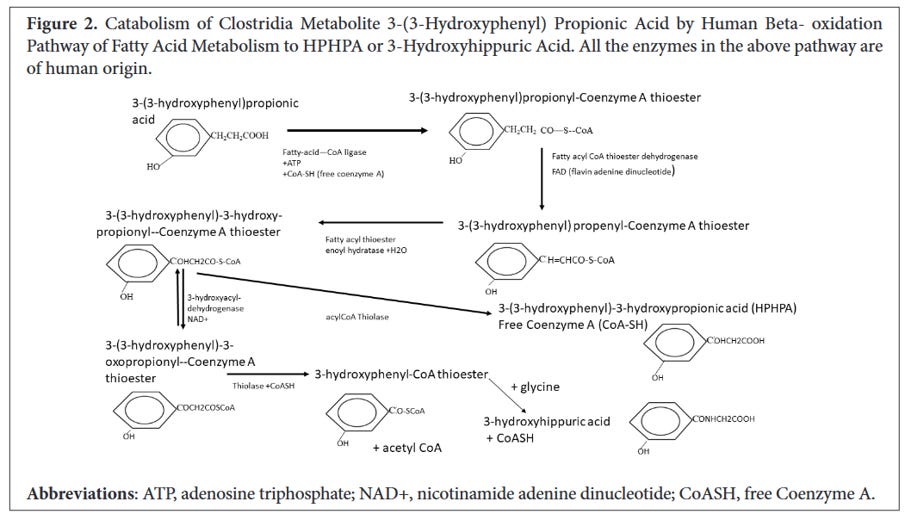
The image below comes from Shaw’s paper and details the steps in the conversion of 3-(3-hydroxyphenyl)propionic acid to HPHPA. The 3-(3-hydroxyphenyl)propionic acid is a phenyl containing fatty acid which fits into the same metabolic pathway for ß-oxidation (discussed below). What’s important to keep in mind is that phenylpropionic acid is a compound produced by various clostridia bacteria such as C. botulinum, C. caloritolerans, and C. sporogenes. It’s this phenylpropionic acid that is the precursor to HPHPA.
Image 1 - this is the chemical structure of phenylpropionoic acid. It is a benzene ring (six hydrocarbon ring structure) with a propionoic acid side chain. Propionoic acid is a three carbon fatty that is typically converted to propionyl-CoA for succinyl-CoA production and entry in the Krebs cycle Propionic acid from intestinal bacteria, and the catabolism of odd chain fatty acids, and amino acids valine and isoleucine are involved in this pathway. Because of its short carbon chain, propionic acid does not enter beta-oxidation.
Image 1 - this diagram from Shaw’s article shows the initial compound 3-(3-hydroxyphenyl)-propionic acid and the various conversion steps to 3-(3-hydroxyphenyl)-3-hydroxypropionic acid (HPHPA). Notice that CoASH is involved in the reaction between 3-(3-hydroxyphenyl)-3-hydroxypropionyl-Coenzyme A thioester to HPHPA, but in other steps within this biochemical chain of events. Source: https://pubmed.ncbi.nlm.nih.gov/37363147/.
Fatty Acid Metabolism and Palmitic Acid
Fatty acid metabolism consists of various reaction steps that take lipid compounds for either the generation of adenosine triphosphate (ATP), or transforms them into building blocks for other compounds. Each metabolic process has its unique purpose for the utilization of lipids in these catabolic and anabolic reactions, respectively. My focus in this article is the catabolic pathway of ß-oxidation which plays a major role in mitochondrial conversion of certain chain-length fatty acids for abundant ATP production. Essentially, ß-oxidation takes fatty acids and converts them into acetyl-CoA. The acetyl-CoA then enters the Krebs cycle generating NADH and FADH2 which carry electrons to the complex I and complex II of the electron transport chain (ETC). Typically, because of the amount of NADH and FADH2 produced and associated electrons that are transferred through the ETC, the amount of ATP produced from fatty acid metabolism greatly exceeds that of glucose.
The ß-oxidation name is derived from the fact that the catabolic reaction starts at the beta (ß) carbon on the fatty acid chain, and then subsequent shortening of the chain occurs in a repeating cycle until the fatty acid has been converted to acetyl-CoA. This reaction primarily occurs within a protein complex associated with the inner mitochondrial membrane.
Image 2 - this is a palmitic acid, the 16-carbon saturated fatty acid that is metabolized through beta-oxidation. CoASH is necessary for this metabolism. It is the most common saturated fatty acid found in animals, plants, and various microorganisms. The palmitate-CoA derivative, along with cholesterol, is need to make activated sonic hedgehog as referenced in article 1.
Fatty Acid Classification
Fatty acids come in various lengths and shapes. They are classified by length (how many carbons in the chain), saturation status (saturated having no double bonds between carbons and unsaturated having one or more double bonds between carbons), even or odd carbon numbers, and whether the structure is linear or branched. Palmitic acid is a saturated, linear, and long-chain fatty acid (LCFA). LCFA’s are typically 13 to 21 carbons long.
All fatty acids have a COOH grouping at one end called a carboxyl group. This functional group provides its acid character with a single carbon double bonded to oxygen and single bonded to a hydroxyl group (-OH). As stated previously, the process of ß-oxidation begins at the beta-carbon. The next image shows the Greek letter designation for carbons distal to the carboxyl carbon.
Image 3 - the Greek lettering begins at the second carbon position with carbon one being part of the carboxyl group. The beta (ß) carbon is two carbons from the carboxyl group, and the gamma carbon is three away from the carboxyl-group.
Beta-Oxidation
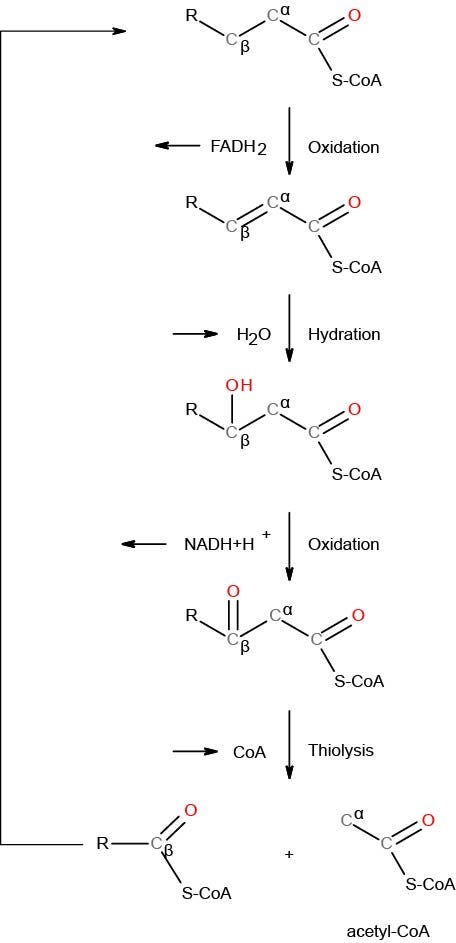
ß-oxidation begins with a dehydrogenation reaction (removes a hydrogen) and forms a double bond between carbon 2 (C2) and carbon 3 (C3). FAD+ acts as an electron acceptor and receiving the hydrogens to become FADH2.
Water is added with a hydroxyl (-OH) group bonding to the ß-carbon (C3). NAD+ participates as the electron acceptor along with hydrogen forming NADH.
The compound is then split between the alpha and beta carbons, C2 and C3, respectively, with the addition of coenzyme-A. This reaction releases the first two carbon units, acetyl-CoA and fatty acyl-CoA (minus two carbons).
Image 4 - the process of ß-oxidation begins with a dehydrogenation reaction and ends with thiolysis. Thiolysis involves the addition of CoASH to one of the products. In this case, the addition of CoASH produces acetyl-CoA and a remaining acyl fatty with two less carbons. The process of this fatty acid metabolism will continue until all the carbons have been turned into acetyl-CoA.
Omega Oxidation
Another fatty acid oxidation reaction is called omega-oxidation. This process is considered a ‘back-up’ reaction if ß-oxidation is compromised from genetic mutations or ß-oxidation cofactor deficiencies such as L-carnitine or CoASH. Excessive substrate into mitochondria such as a high fat diet can also activate the omega-oxidation pathway. An organic acid compound called adipic acid can be elevated when the ß-oxidation pathway is faulty. The image below is adipic acid which is a 6-carbon dicarboxylic acid. Omega-oxidation does not require CoASH as a cofactor.
Image 4 - adipic acid is a product of omega oxidation and can appear when ß-oxidation is compromised. This marker was seen elevated in the Mussap, et al, research group analyzing compounds linked to autism.
In the Mussap, et al research from Italy, the authors showed that palmitic and adipic acids, along with HPHPA were highly diagnostic of autism. It is not uncommon to see high adipic acid on organic acids tests from autistic individuals.
Membrane Transport
Because fatty acids carry negative charges they cannot easily pass through cell membranes. Their entry into the cell is handled by cell membrane transport proteins which delivers them into the cytosol. For transport into the mitochondria where ß-oxidation takes place, the mechanism of transport is dependent on L-carnitine. This is particularly true of long-chain fatty acids like palmitic acid. The mechanism involved in this transport is beyond the scope of this article, but the next image provides a visual representation of the reactions involved to get certain length fatty acids into the mitochondria.
Image 5 - this image shows the relationship between carnitine and acyl-CoA for the transfer of fatty acids (certain chain lengths) across the inner mitochondrial membrane for ß-oxidation activation.
A good take home point with regards to this mechanism, and the general discussion of ß-oxidation fatty acid metabolism disruption and HPHPA, is the use of L-carnitine. This important nutrient does not have a direct treatment effect on clostridia and its production of HPHPA. However, it can provide benefit for improving the efficiency of long-chain fatty acid metabolism, including palmitic acid.
Propionic Acid
The final thing I want to mention in this article as it relates to clostridia bacteria and HPHPA, is that of propionic acid. It has been established that various clostridia bacteria produce phenylpropionic acid from fermentation of phenylalanine. Propionic acid can also come from odd chain fatty acids and some amino acids. Other bacteria in the digestive system can produce propionic acid too as a normal part of its function. There is evidence that this acid may have some modulation influence within the immune system.
Image 6 - propionyl-CoA to succinyl-CoA is dependent on free CoASH just like ß-oxidation.
Conclusion
CoASH, just like in ß-oxidation, is important in propionic acid metabolism too producing propionyl-CoA which is involved in succinyl-CoA production for Krebs cycle activation. The final image above takes us back to article 2 showing the relationship between various compounds, their conversion to propionyl-CoA, and another entry point into the Krebs cycle for metabolic energy production.
In part #4 of this article series I will explain in more detail some of the other problems of HPHPA, as well as 4-cresol from clostridia, and their inhibition of dopamine beta-hydroxylase (DBH) before concluding with a final analysis again of HPHPA, free CoASH, palmitic acid, and sonic hedgehog.